Other hydrophilic amino acids have polar side chains that are basic, like lysine, histidine, and arginine.Īt physiological pH the acidic groups lose a hydrogen and the basic groups gain a hydrogen.įinally, some polar side chains are neutral, for example they can contain hydroxyl groups, -OH, like serine, threonine, or tyrosine, or sulfhydryl groups -SH, like cysteine, or carboxamide groups (R-C=0-NH2) like asparagine or glutamine. These polar side chains might be acidic - like when their side chains contain additional carboxyl -COOH groups, like aspartic acid and glutamic acid. Now, hydrophilic amino acids have polar side chains. This might be in the form of an alkyl side group, which is a saturated hydrocarbon, seen in valine, glycine, alanine, leucine, isoleucine, methionine, and proline.Īlternatively, it might be in the form of an aromatic side group - which involves a 6-carbon ring, like in phenylalanine, tyrosine, and tryptophan. Hydrophobic amino acids have nonpolar side chains. Now, the alpha carbon also has a side chain, sometimes marked as “R”.Īnd this side chain gives the amino acid certain properties, which can play an important role in the overall protein structure.įirst the side chain can be hydrophilic or hydrophobic - so water loving or water hating. Having both a positive and a negative charge makes amino acids a type of zwitterion - which is German for “hybrid”, or “double ion”. Now, at a physiologic pH of 7.4, the amine group has a positive electrical charge, and the carboxyl group has a negative charge. The amine and carboxylic acid groups are both bound to the same carbon, called the alpha carbon. Just from the name, you can tell they’ve got an amine group, or “NH2”, and also an acid, in this case a carboxylic acid group “COOH”. Then, there’s 6 of them that we call conditionally essential because we can make them most of the time, but not always - arginine, cysteine, glutamine, glycine, proline, and tyrosine.įinally, there are 9 of them that we cannot make ourselves - His, Ile, Leu, Lys, Met, Phe, Thr, Trp, and Val, and as a result we have to obtain them from our diet. There are 5 amino acids that are dispensable - alanine, aspartic acid, asparagine, glutamic acid, and serine - because we can make them de novo ourselves at any time, and in good quantity.

One way to divide them, is into the ones that we make ourselves, and the ones that we cannot.
So, we have: alanine (Ala), arginine (Arg), asparagine (Asn), aspartic acid (Asp), cysteine (Cys), glutamic acid (Glu), glutamine (Gln), glycine (Gly), histidine (His), isoleucine (Ile), leucine (Leu), lysine (Lys), methionine (Met), phenylalanine (Phe), proline (Pro), serine (Ser), threonine (Thr), tryptophan (Trp), tyrosine (Tyr), valine (Val). Humans use 20 amino acids in our day-to-day protein making.

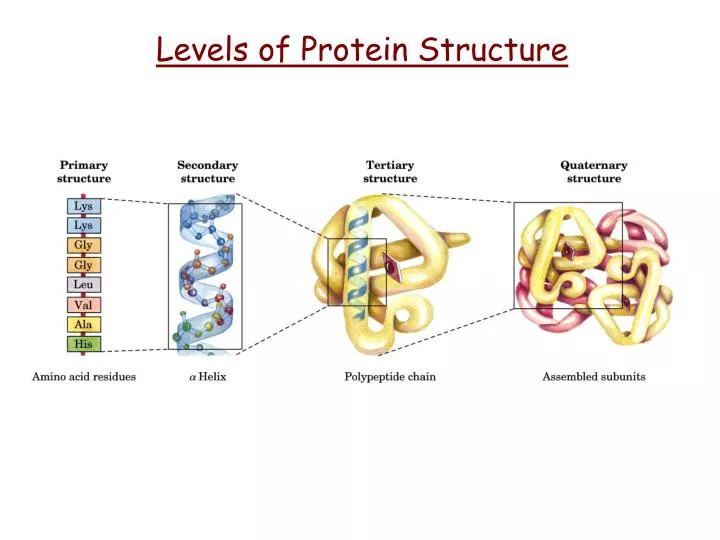
To make a protein, we need to get to know two things - the “ingredients”, which are the amino acids, and the “recipe” - or how the finished amino acid chain folds into the protein. They sorta resemble a string of beads, and they get twisted and folded into a final protein shape. Proteins are vital for the normal function of a cell.Įssentially, a protein is, at its simplest, a very long chain of individual units, called amino acids, bound to each other by peptide bonds to form an amino acid chain.
